 Nitrogen, N 2 (g) – see CLEAPSS Hazcard HC068A.Oxygen, O 2 (g), (OXIDISING) – see CLEAPSS Hazcard HC069.Hydrogen, H 2 (g), (EXTREMELY FLAMMABLE) – see CLEAPSS Hazcard HC048.Remove all possible sources of ignition when handling flammable gases.Read our standard health and safety guidance.The experiment requires gas bags filled with various gases: 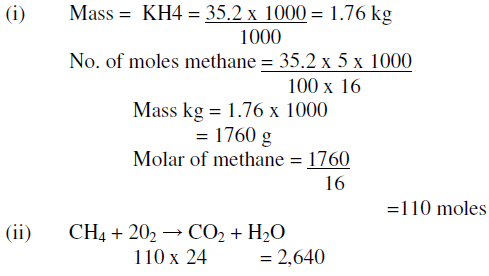
Access to an accurate measurement of atmospheric pressure.Thermometer to measure room temperature, accurate to +/– 0.5 ✬, with digital display if available.Top-pan balance, accurate to +/– 0.001 g, with output to a computer to display readings, if available (an accuracy of +/– 0.01 g is not really sufficient for successful results).Cable tie (to fit around the rubber bung).Rubber septum cap, self-sealing, to fit the glass tubing.Glass tubing, 4–5 cm length, to fit rubber bung.Rubber bung, one hole, 17 mm or similar.Polythene bag (sandwich or freezer bag), 1–2 dm 3 capacity.For each gas bag (see notes on creating the gas bags below):.Small rubber bung (exact size unimportant).Syringe (disposable plastic), 50 cm 3 or larger if available, fitted with hypodermic needle (see notes on preparing the syringe below).The demonstration itself should then take about 10 minutes for each gas used. 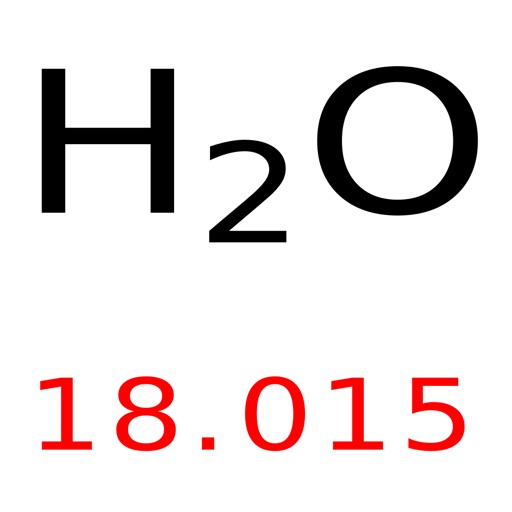
The preparation of bags containing different gases, and the modification of the syringes, should be done before the lesson, as these procedures may take a considerable time. Teachers of advanced students may wish to consider the possibility of a student practical for students with good manipulative skills, using appropriate gases. This is most likely to be done as a teacher demonstration. In this demonstration, the relative molecular masses of various gases are determined by weighing containers before and after they are used to collect the gases RSC Yusuf Hamied Inspirational Science Programme. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
